
When this compound comes in contact with water or moist air, it is decomposed and phosphine (PH 3) is formed. 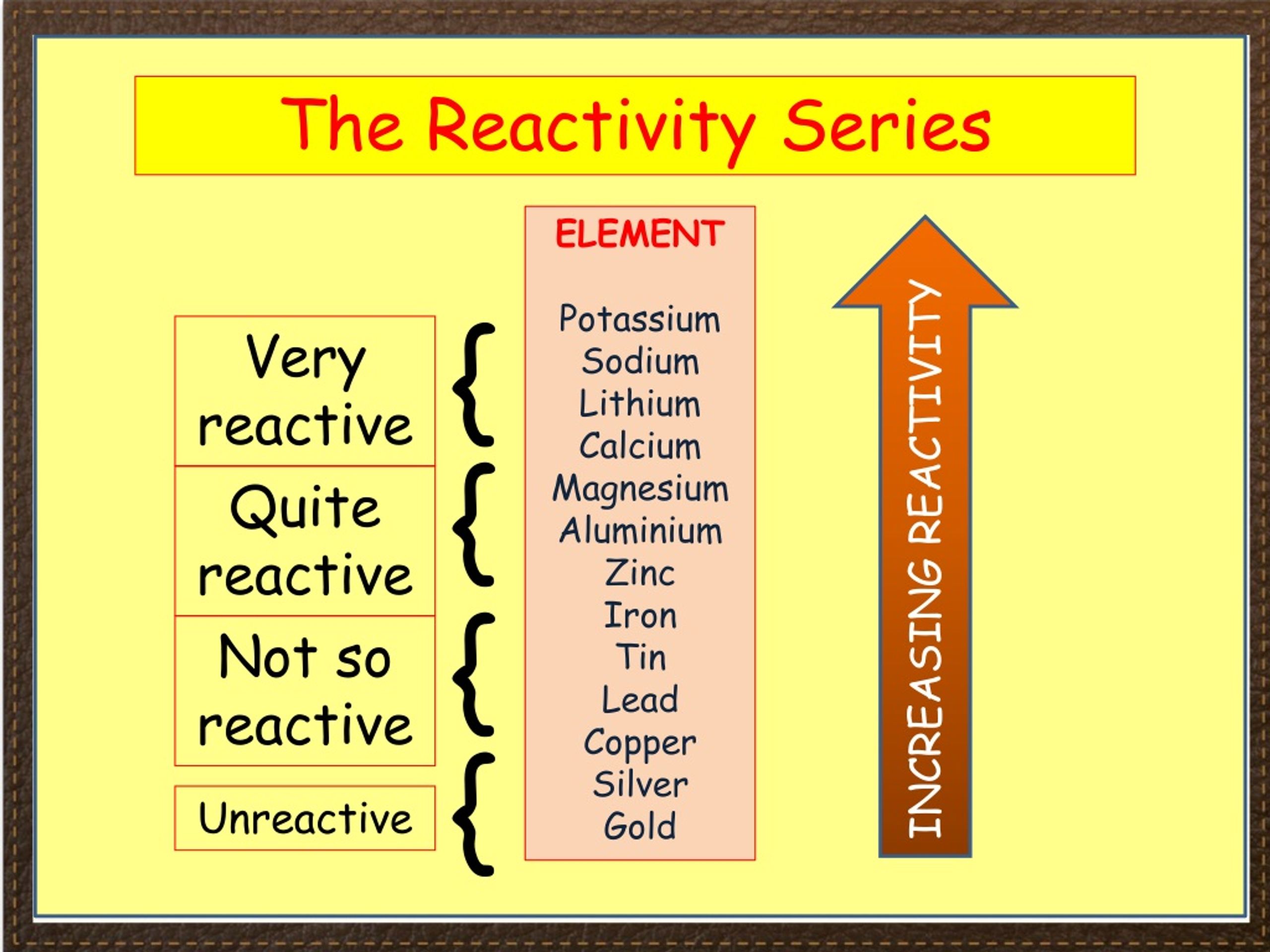
To prevent any damage, a magnesium fire must be covered in sand.Īn example of a magnesium compound is magnesium phosphide (Mg 3P 2), an odorous, grey solid. When attempts are made to extinguish magnesium fires with water, magnesium aggressively reacts with hydrogen gas. It than reacts with nitrogen from air to form magnesium nitride (Mg 3N 2). Magnesium continues to burn after oxygen is depleted. Magnesium fires cannot be extinguished by water. Furthermore, magnesium reacts with water vapor to magnesium hydroxide and hydrogen gas: 
Magnesium generally is a slow-reacting element, but reactivity increases with oxygen levels. Magnesium metals are not affected by water at room temperature. In what way and in what form does magnesium react with water? Water containing large amounts of alkali earth ions is called hard water, and water containing low amounts of these ions is called soft water. Magnesium and other alkali earth metals are responsible for water hardness. Rivers contains approximately 4 ppm of magnesium, marine algae 6000-20,000 ppm, and oysters 1200 ppm.ĭutch drinking water contains between 1 and 5 mg of magnesium per liter. After sodium, it is the most commonly found cation in oceans. Magnesium (Mg) and water Magnesium and water: reaction mechanisms, environmental impact and health effects Magnesium is present in seawater in amounts of about 1300 ppm.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
